Combination Ultrasound Technique Treats Ureteral Stones
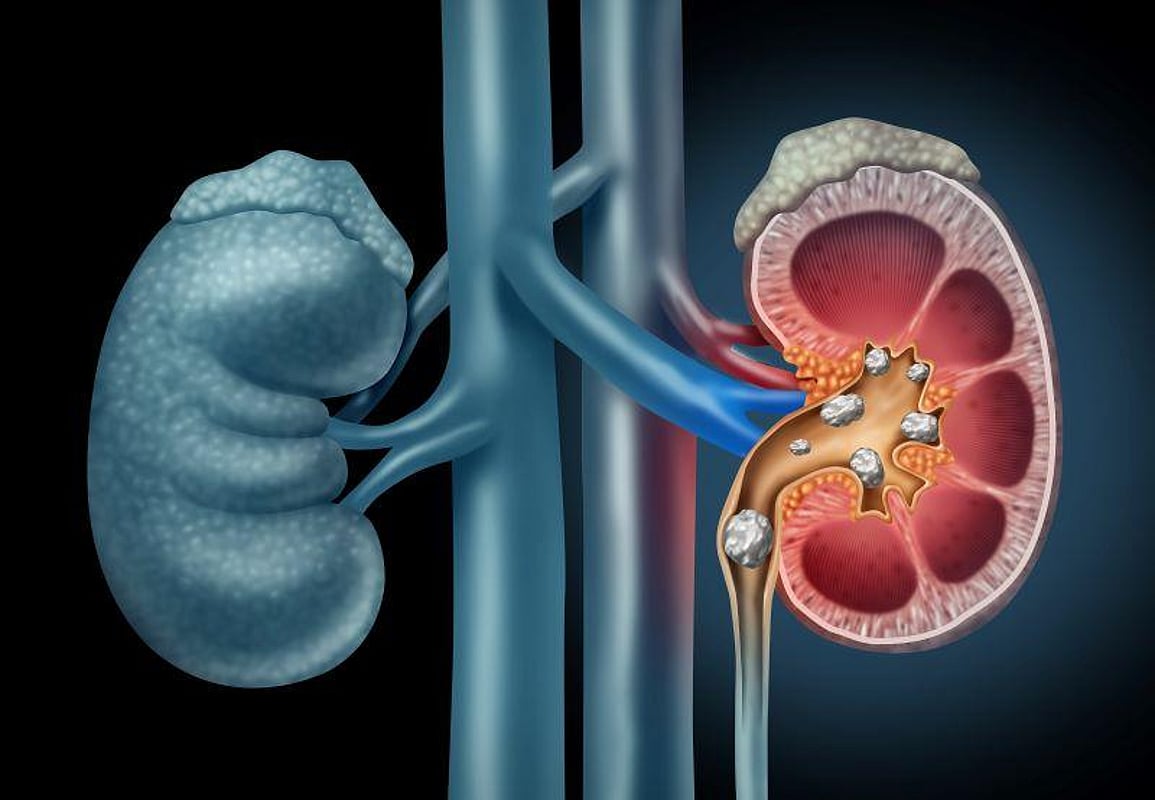
TUESDAY, Oct. 18, 2022 (HealthDay News) -- The combination of ultrasonic propulsion and burst wave lithotripsy can be used in awake patients to reposition and break up ureteral stones to facilitate passage, according to a study published in the November issue of The Journal of Urology.
M. Kennedy Hall, M.D., from the University of Washington School of Medicine in Seattle, and colleagues tested transcutaneous focused ultrasound in the form of ultrasonic propulsion and burst wave lithotripsy to reposition ureteral stones and facilitate passage in awake patients. The analysis included 29 individuals receiving either ultrasonic propulsion alone (16 patients) or with burst wave lithotripsy bursts (13 patients).
The researchers found stone motion in 66 percent of patients. In an average of 3.9 days after the procedure, the stone passed in 18 of the 21 distal ureteral stone cases with at least two weeks of follow-up. In seven of 13 burst wave lithotripsy cases, fragmentation was seen. The procedure was tolerated by all. Hematuria on initial urination postprocedure and mild pain were anticipated. Seven participants had associated discomfort, with only 18 of 820 propulsion bursts (2.2 percent).
“This technology has the potential to provide an advantageous and patient-centered solution for the gap between expectant management and urological intervention for symptomatic ureteral stones,” the authors write.
Several authors disclosed financial ties to SonoMotion, which has licensed this technology from the University of Washington for commercialization.
Related Posts
Your Guide to Pain Relief After Knee, Hip Replacement
FRIDAY, Oct. 21, 2022 (HealthDay News) -- Getting a new knee or hip can be...
Un estudio nuevo halló que la mayoría de las personas infectadas con Ómicron pueden no saberlo
Was this page helpful?
Lipid-Lowering, Antidiabetic Drugs Tied to Prevalence of AMD
TUESDAY, Nov. 8, 2022 (HealthDay News) -- Lipid-lowering drugs (LLDs) and...
FDA Panel Skeptical of Controversial ALS Drug Ahead of Vote
TUESDAY, Sept. 6, 2022 (HealthDay News) – A U.S. Food and Drug Administration...
